Quality control for hydrolyzed diets for cats and dogs
Written by Jérôme Naar and Isabelle Lesponne
Identifying a suitable diet for pets with adverse food reaction can be less than straightforward and pitfalls await the unwary; here Jérôme Naar and Isabelle Lesponne outline how Royal Canin ensure their Anallergenic diets are free from unwanted proteins.
Article

Key Points
Recent studies have highlighted that the contents of many diets claiming to be suitable for adverse food reactions are potentially not fit for purpose.
Royal Canin Anallergenic diets are manufactured to exacting standards which ensure they are appropriate for animals that suffer from adverse food reactions.
Adverse Food Reactions (AFR) in dogs and cats are a not-uncommon cause of dermatological signs presented in clinical practice. Commercial AFR diets are often used in both the diagnosis (via an elimination trial) and treatment of the condition, and two broad categories of diets are available:
1. “Selected“ or “novel“ protein diets, with limited protein sources and – ideally – a single carbohydrate source.
2. “Hydrolyzed diets“, consisting of partially or extensively hydrolyzed protein.
Elimination diet trials can be subject to various potential failures. Two major concerns are that the AFR diet must be free of ancillary proteins (as potential cross-contamination in petfood factories is a major concern) and (for hydrolyzed diets) have a guaranteed level of hydrolysis, with no remaining allergenic fragments present. Recent studies have highlighted that some commercial (mostly “over-the-counter”) diets marketed for AFR contain undeclared ingredients and/or large molecular weight proteins; for instance, discrepancies were found between protein analyses and labeling in around 75% of diets tested in recent studies [1] [2] [3]. Consequently, to ensure the absence of cross-contaminated products, three key elements are required: fully characterized raw materials, fit-for-purpose industrial equipment and cleaning processes, and optimal analytical controls.
A recent study sought to confirm the specific protein composition of Royal Canin’s Anallergenic canine and feline diets (Ultamino in North America) – which are based on extensively hydrolyzed poultry feather and cornstarch – and the efficacy of the cross-contamination risk management process.
• Protein composition was assessed via high-sensitivity chromatographic, electrophoretic and proteomic techniques by an external independent laboratory. The results obtained confirmed the extensive level of protein hydrolysis (Figure 1), the absence of protein in the palatability enhancers (included in the kibble coating), and the absence of cross-contamination [4]. The ubiquitous corn starch glycoprotein GBSS (granule-bound starch synthase) was detected, which to date has no known adverse effects in pets, but lipo-transfer proteins (LTPs) – which are well-recognized to be allergenic – were not detected. The same results were obtained for both canine and feline diets [4].
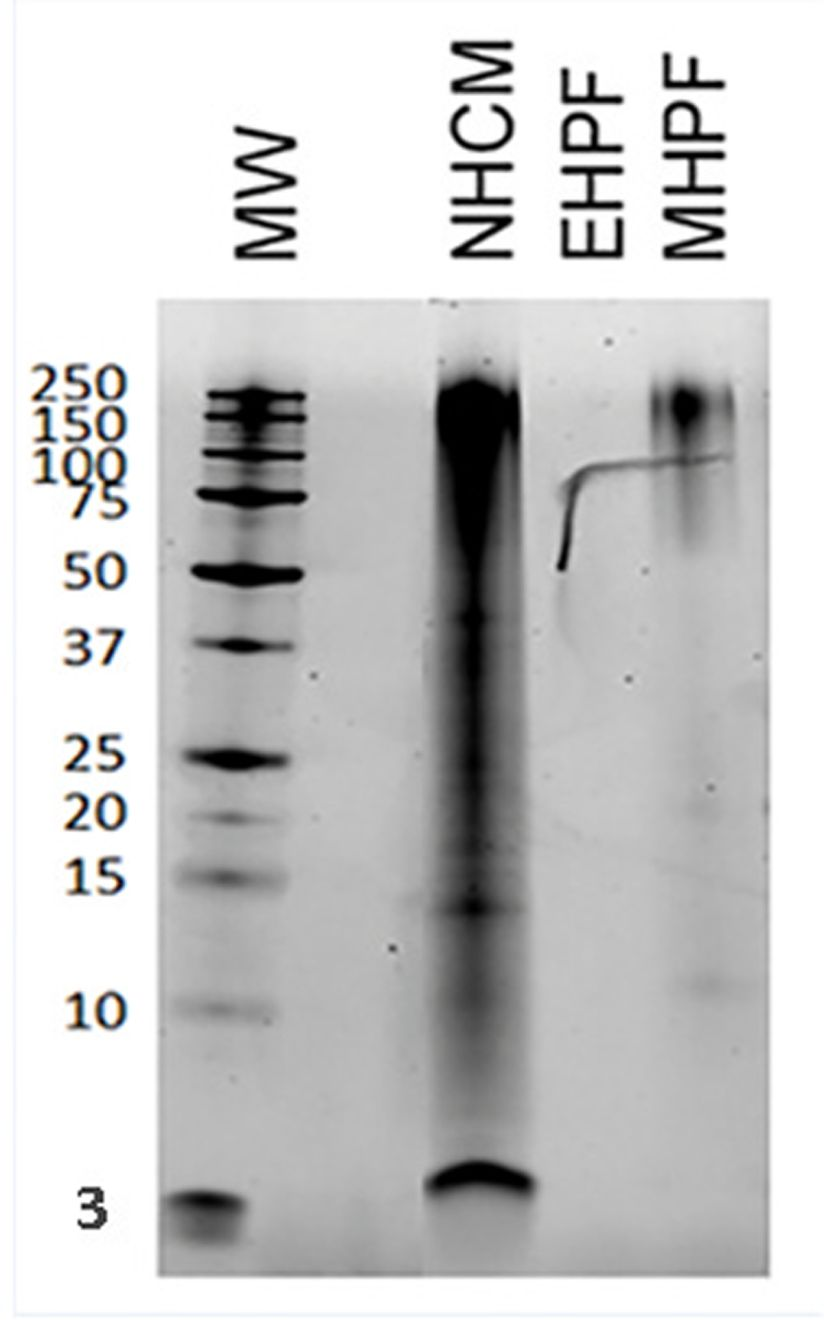
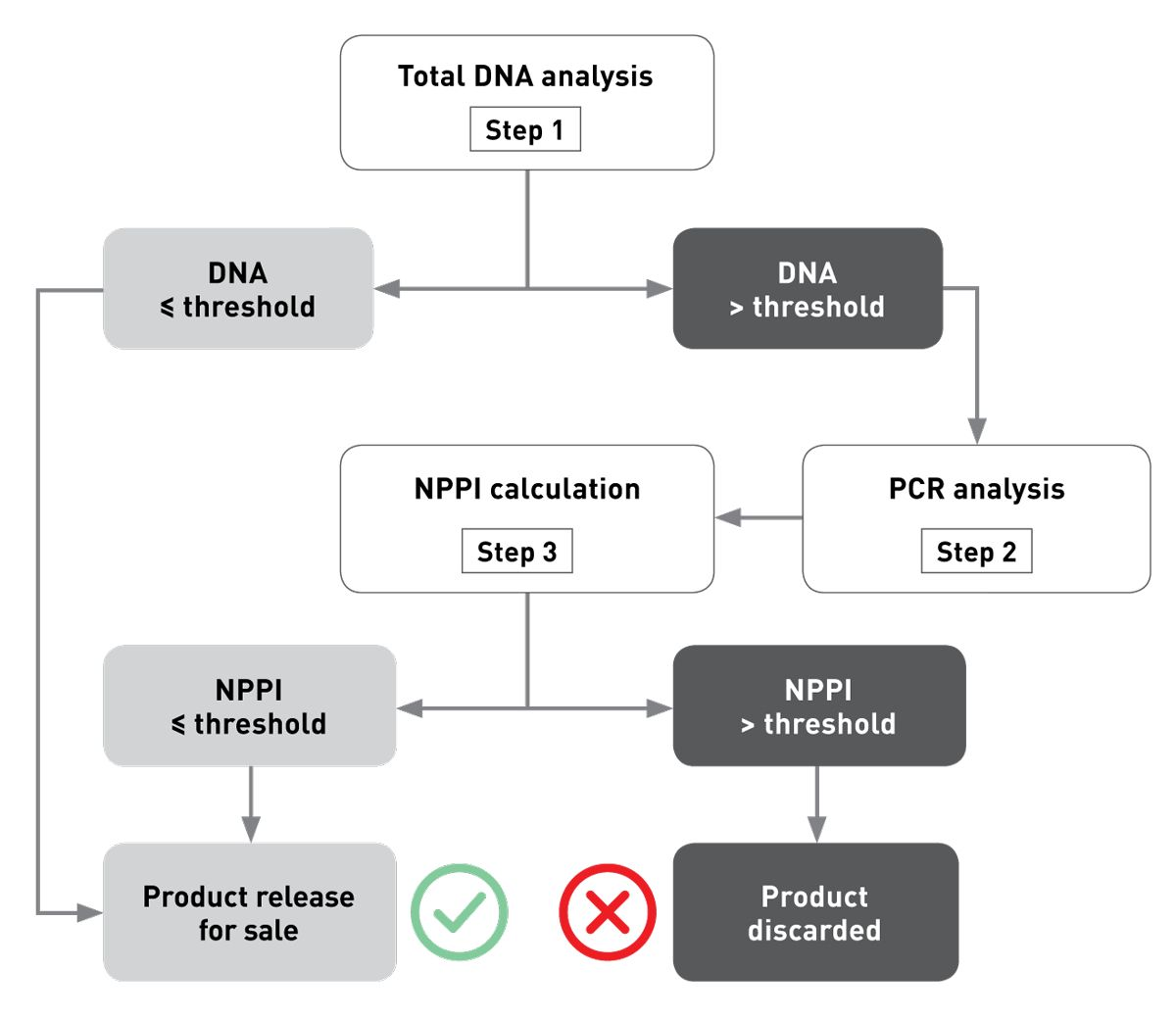
• Cross-contamination risks are assessed by testing each production batch using a specific 3-step DNA-based analysis (Figure 2). This involves measurement of the total DNA content of the diet and comparing the results with a conformity threshold established during a multicenter pre-clinical trial [5]. If DNA is detected above this threshold, PCR analyses are conducted to identify the contamination source. To correlate the DNA to the unwanted proteins, “calibration curves” are then employed to assess the actual concentrations of protein present.
Since launching in 2011, this method has been employed to test more than 2,500 production batches of Anallergenic, with all found to conform to the required standards. This has allowed the batches to be released from the factory for sale[6] [7] [8].
Acknowledgements to Mars Petcare Central Laboratory, Aimargues (France) and the Luxembourg Institute of Science and Technology.
Because of the strict quality control features (careful selection of raw materials, scrupulous factory protocols and cleaning methods, and rigorous DNA tests), no cross-contamination is present in these diets which, along with proven extensive protein hydrolysis, ensures optimal clinical reliability and efficacy for using Anallergenic in AFR diagnosis and management.
Jérôme Naar
PhD
France
Dr. Naar has a background in biochemistry, environmental science and toxicology and joined Royal Canin in 2012 as an expert within the Quality & Food Safety team in R&D. Prior to this, he worked with the Pasteur Institute in French Polynesia before relocating to the USA to manage a research laboratory specializing in environmental toxicology.
Isabelle Lesponne
DVM
France
Dr. Lesponne graduated from the National Veterinary School of Toulouse in 2001 and worked in small animal practice for several years before moving to an animal pharmaceutical company in 2007. She has served as scientific support manager at Royal Canin R&D since 2011.
References
- Horvath-Ungerboeck C, Widmann K, Handl S. Detection of DNA from undeclared animal species in commercial elimination diets for dogs using PCR. Vet Dermatol 2017;28(4):373-e86.
- Ricci R, Granato A, Vascellari M, et al. Identification of undeclared sources of animal origin in canine dry foods used in dietary elimination trials. J Anim Physiol Anim Nutr (Berl) 2013;97 Suppl 1:32-38.
- Roitel O, Maurice D, Douchin G, et al. High molecular weight proteins in hydrolysed dog foods. Vet Dermatol 2015;26:304.
- Lesponne I, Naar J, Montano M, et al. DNA and protein analyses support the clinical reliability of an extensively hydrolysed diet. Vet Dermatol 2017;28:11.
- Mougeot I, Weese H, Sauve S, et al. Clinical efficacy of a highly hydrolyzed poultry feather protein-based diet for canine AFR diagnosis and dietary management: a 12 case pilot study. In Proceedings, Waltham International Nutrition Sciences Symposium 2013.
- Bizikova P, Olivry T. A randomized, double-blinded crossover trial testing the benefit of two hydrolysed poultry-based commercial diets for dogs with spontaneous pruritic chicken allergy. Vet Dermatol 2016;27(4):289-e70.
- Boutigny L, Lesponne I, Feugier A, et al. Evaluation of a new extensively hydrolyzed poultry feather protein-based diet for the dietary management of feline adverse food reaction (AFR): a 15 cases pilot study. Poster, SEVC congress, Barcelona, 2017.
- Cadiergues MC, Muller A, Bensignor E, et al. Diagnostic value of home-cooked and an extensively hydrolysed diet (Anallergenic, Royal Canin, France) in the diagnosis of canine adverse food reaction: a randomized prospective multicenter study in 72 dogs. In: Proceedings, World Veterinary Dermatology Congress 2016.
Other articles in this issue
Share on social media